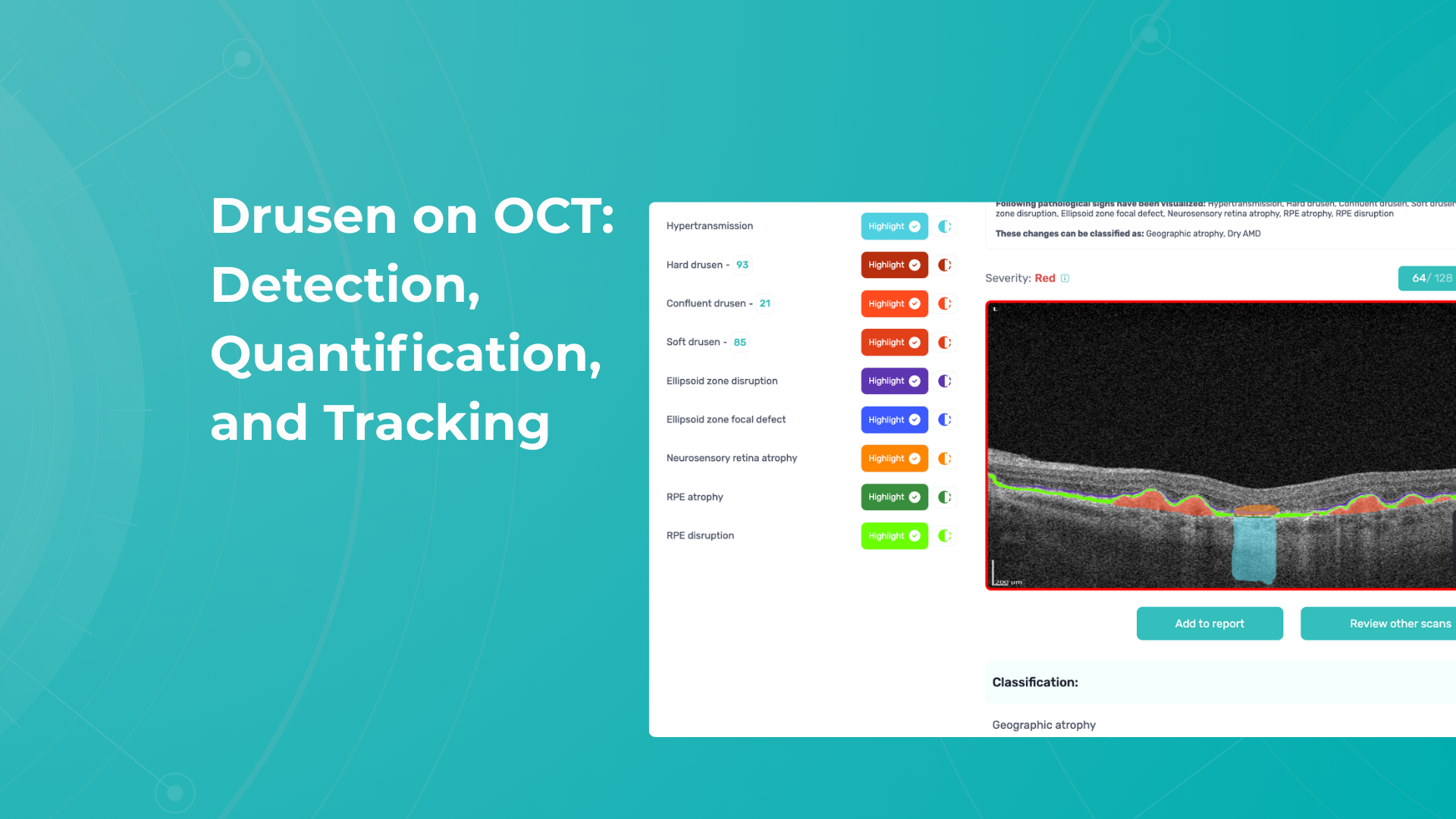
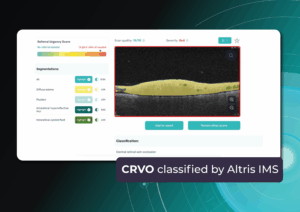
Recently Posted
-

AI Blindness Prevention: how AI can combat vision loss?
 Maria Martynova
07.08.20239 min read
Maria Martynova
07.08.20239 min readAI for Eye Diseases: how AI can combat vision loss?
The total number of people with near or distant vision impairment reaches 2.2 billion worldwide.
Of these, 43 million people are blind, and 295 million are suffering from moderate to severe visual impairment. Although the numbers are constantly changing as new research is conducted, the global burden of blindness and visual impairment remains a significant problem of humanity in the fight against which specialists combine their forces with AI technologies.

AI blindness prevention tools are being actively developed to transform the landscape of vision care in many ways. Eye care specialists use AI systems for screening and detecting diseases that lead to vision loss. AI-powered smart monitors assist specialists in finding proper contact lenses and glasses. In addition, many researches are held with the help of AI algorithms, as they are able to process vast amounts of data.
In this article, we will discuss different applications of AI in blindness prevention, specifically how artificial intelligence tools can empower eye care specialists and extend beyond the clinical setting.
How to Prevent Blindness: Conditions and Risk Factors
Before talking about the developments in the AI sector toward blindness prevention, we would like to discuss the most common causes and risk factors of this impairment. Many health and lifestyle factors can influence the risk of vision loss. Smoking, excessive alcohol consumption, sun exposure, and poor nutrition can contribute to diseases that lead to vision loss.
In addition, there are many conditions that can lead to blindness if left with no proper treatment, among which are the following.
Age-related eye diseases
The global population is aging rapidly. The number of people aged 65 and over is projected to triple from 1 billion in 2020 to 2.1 billion in 2050. Considering this fact, age-related eye diseases have become a prominent cause of blindness. Such diseases as age-related macular degeneration (AMD), cataract, and glaucoma are more prevalent in older patients, and if left untreated, they can lead to fast and significant vision loss. Regular eye check-ups and timely interventions are crucial in managing these diseases and preventing severe visual impairment.

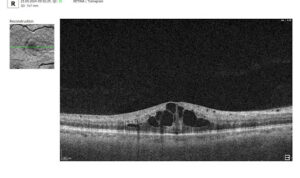
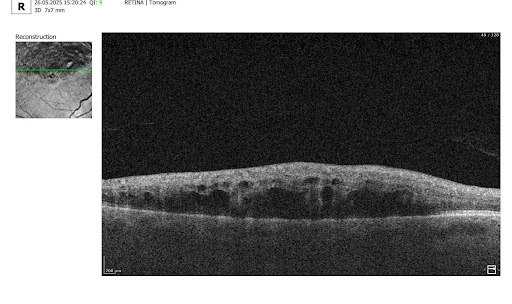
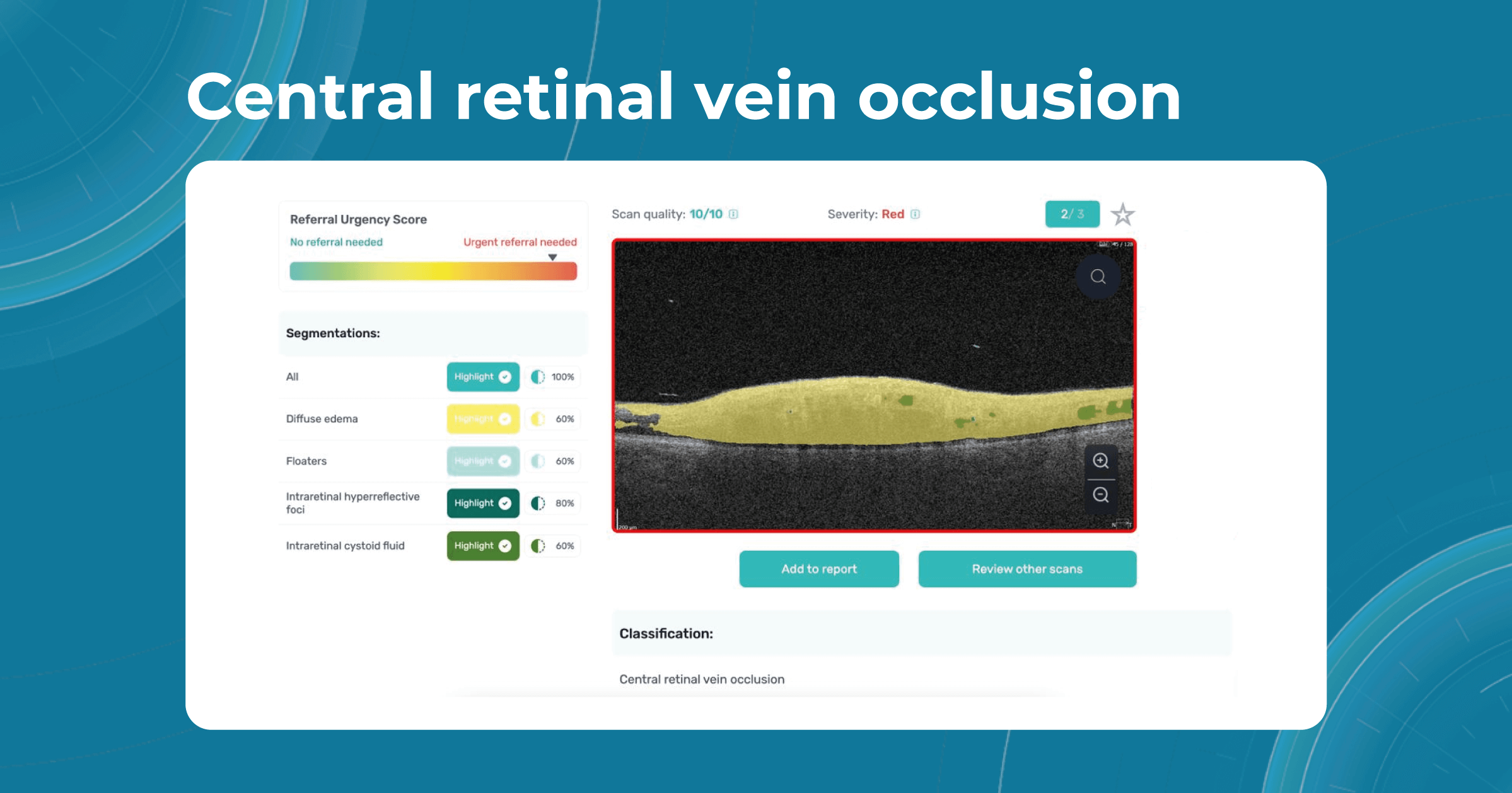
Besides AMD, there are a lot of age-related conditions which can be a red flag when examining the patient. Among these are macular holes, mactel, and vascular diseases, for example, central retinal vein occlusion (CRVO) and central retinal artery occlusion (CRAO). Detecting even one of these pathological conditions in the early stages of their development is crucial for preventing vision loss.
However, many eye care specialists sometimes don’t have enough resources to dedicate more time to analyzing patients’ images. Our recent survey detected that among more than 300 participating optometrists, 40% of them have more than 10 OCT exams per day. Meanwhile, 35% of eye care specialists have 5-10 OCT examinations per day. The greater the number of patients per day, the greater the likelihood that eye care specialists may miss some minor, rare, or early conditions.

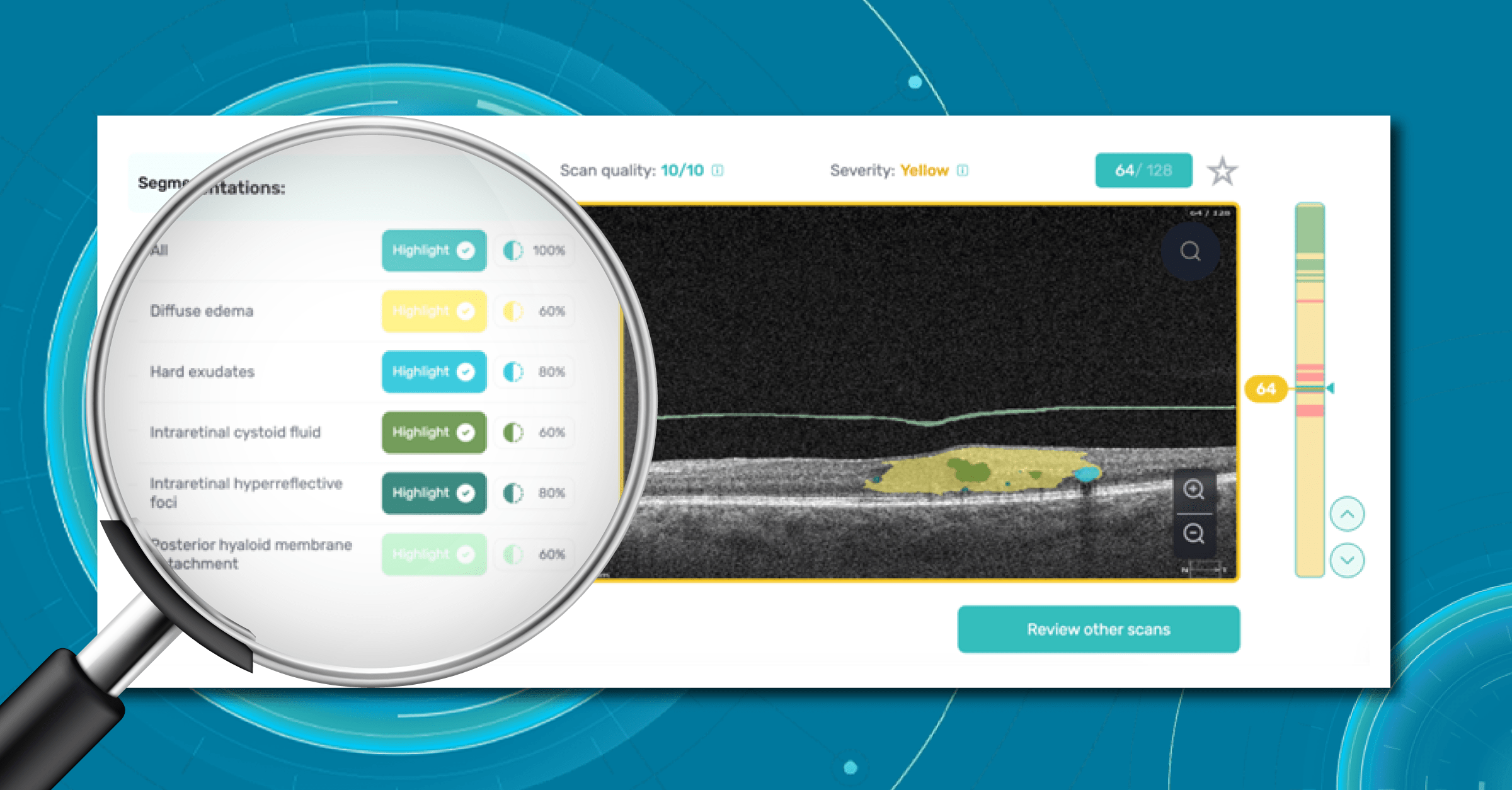
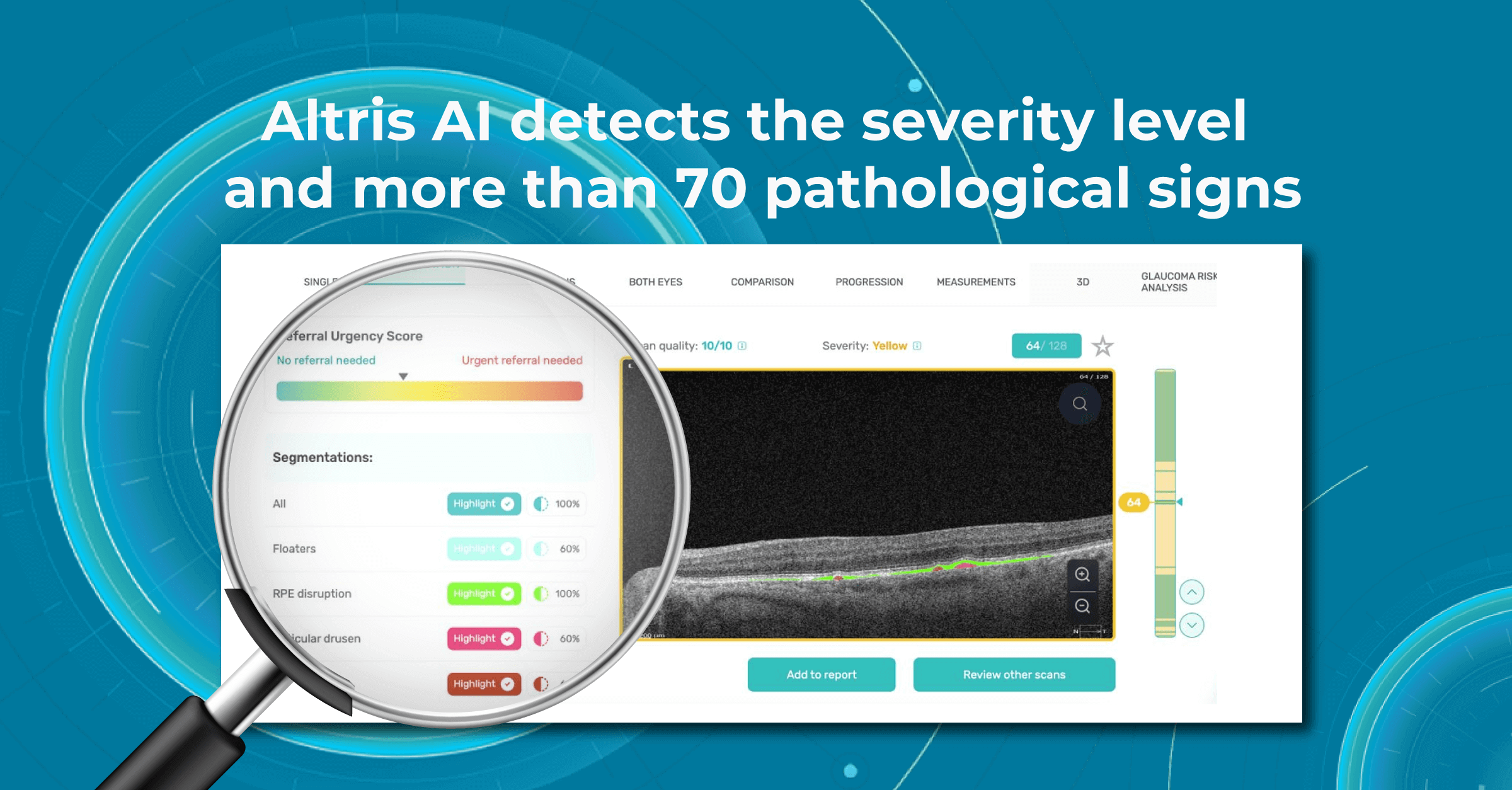
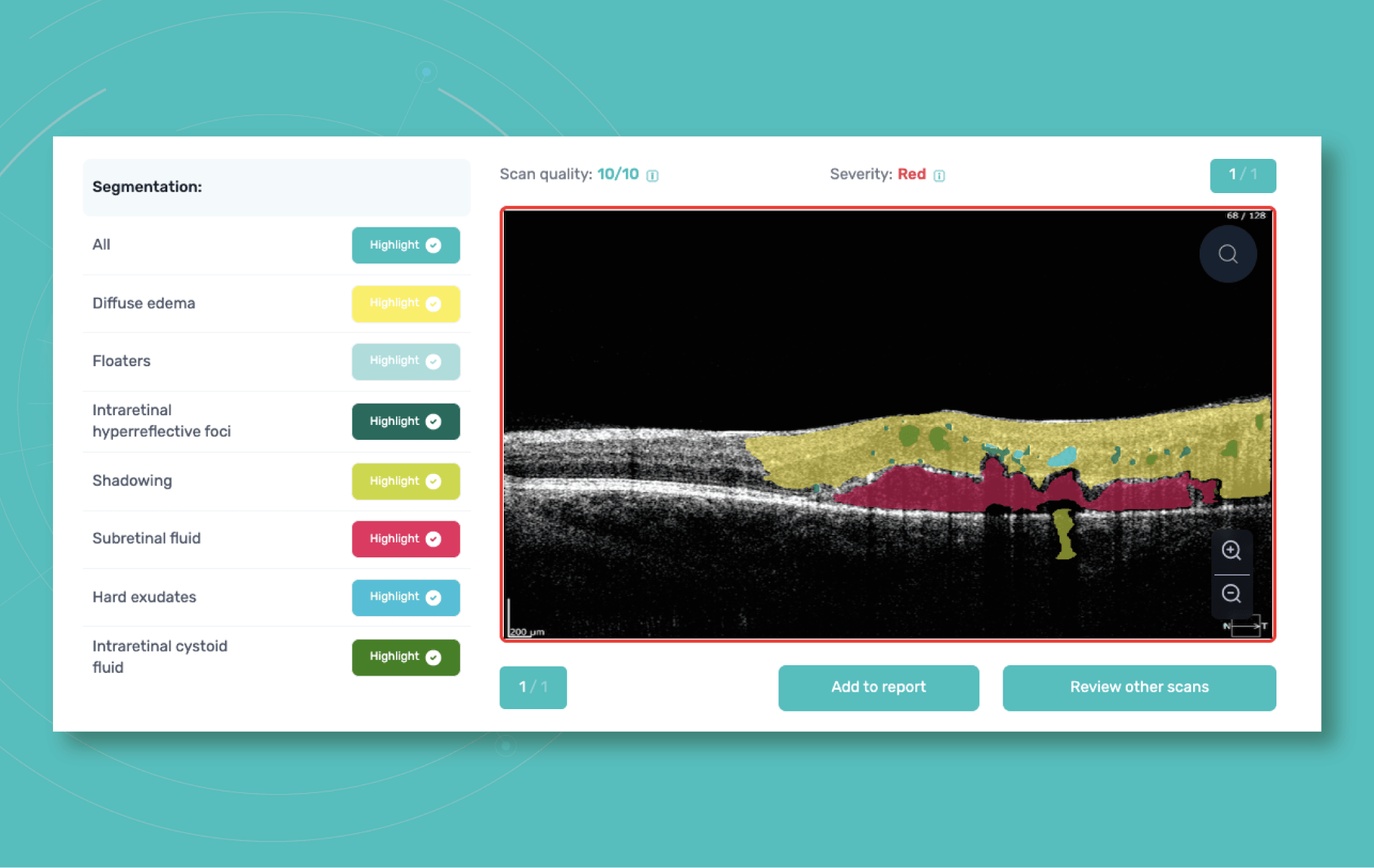
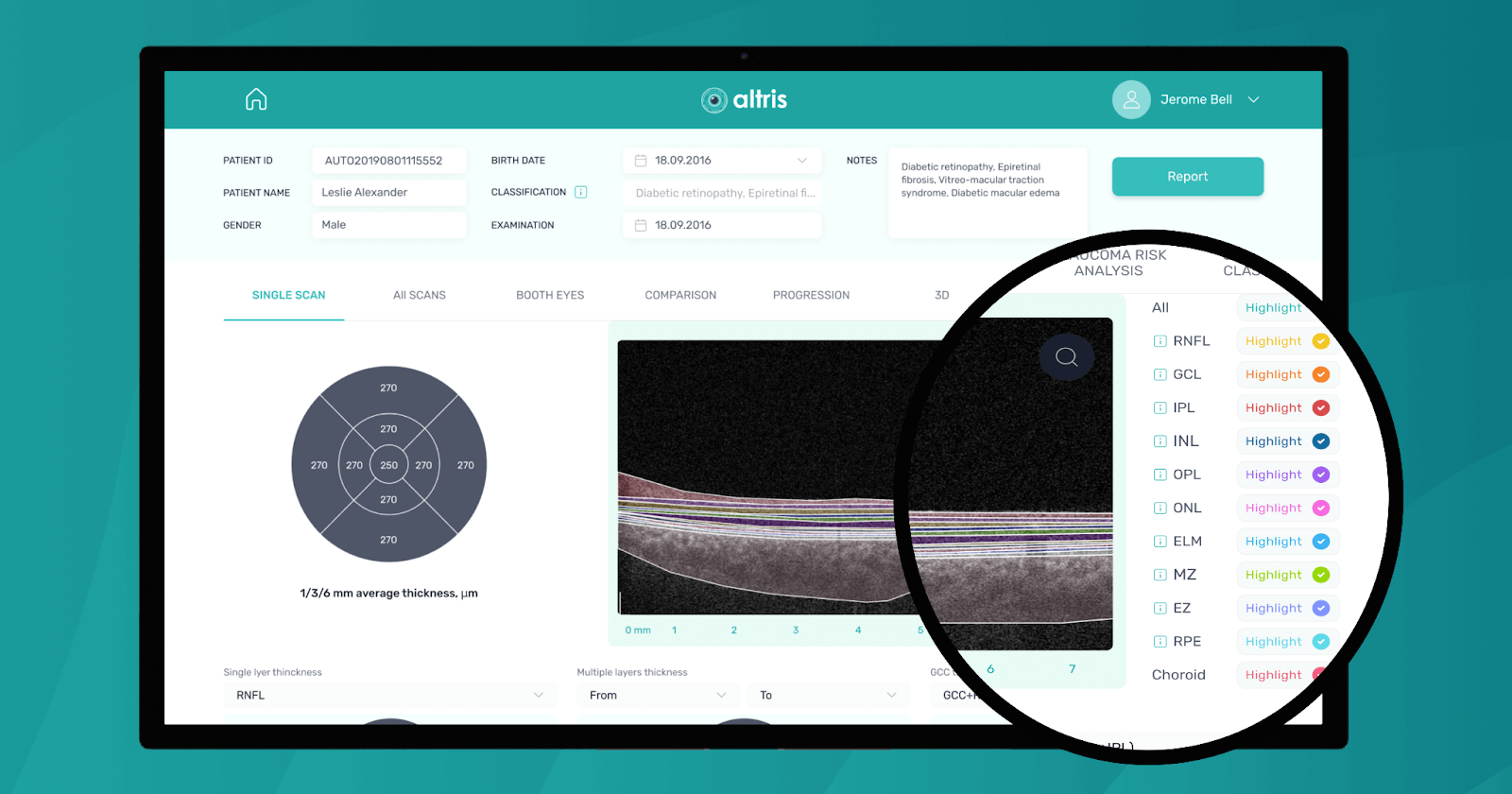
Fortunately, nowadays, there are a lot of ways to empower the clinical workflow, and AI blindness prevention tools are gaining popularity. Artificial intelligence systems like Altris AI can analyze retinal images and other diagnostic data to detect early signs of age-related eye diseases. Altris AI platform, for example, can detect 70+ pathologies and pathological signs, including the ones, that refer to age-related diseases.

Altris AI platform allows eye care specialists to rely on its disease classification when diagnosing a patient. It detects all the most common age-related pathologies, such as AMD, mactel, and vascular diseases – CRVO, CRAO.

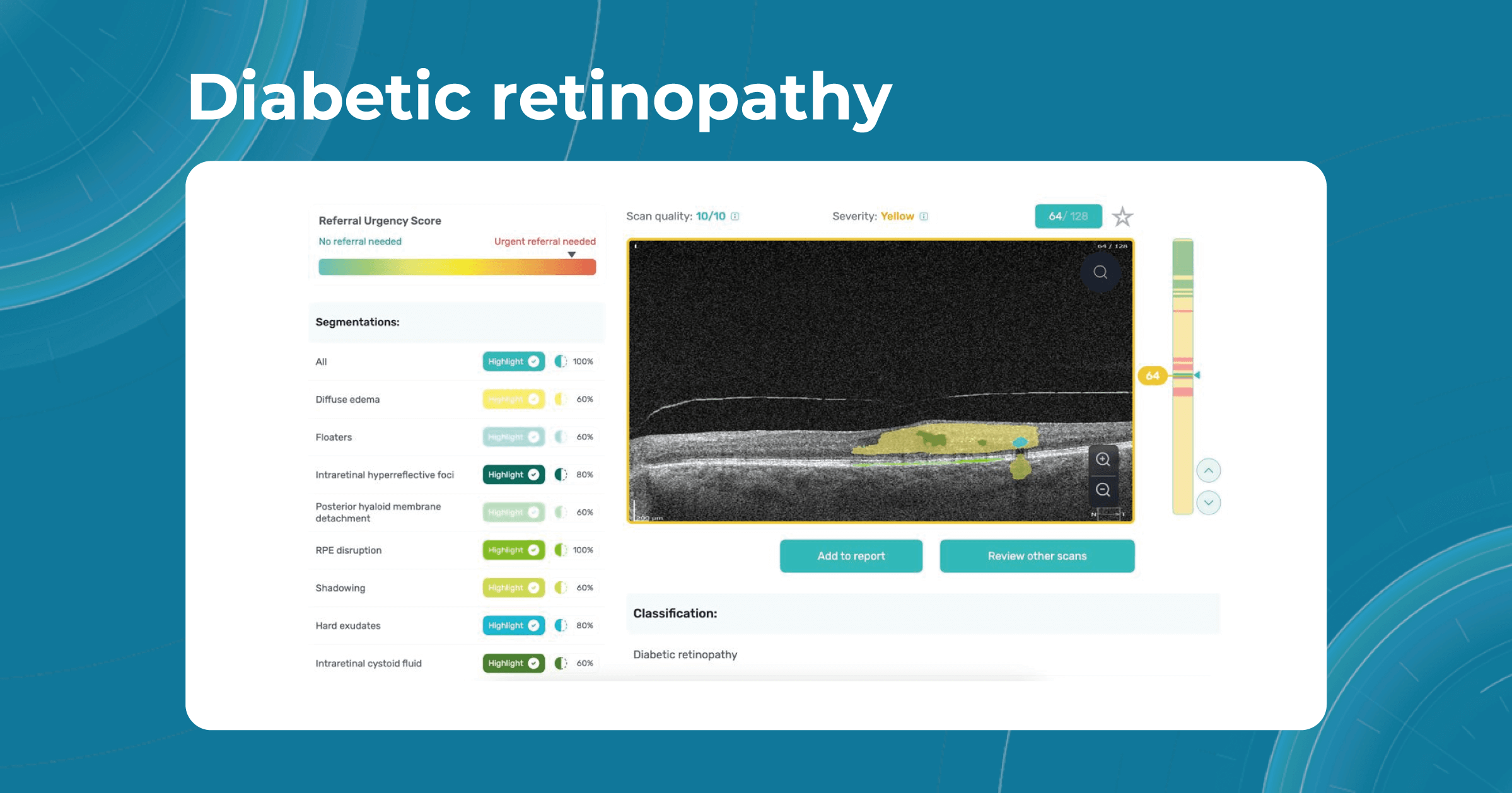
Diabetes and diabetic retinopathy
Diabetes and related conditions are also common causes of vision loss. In the United States, about 12% of all new cases of blindness are caused due to diabetes. Globally, diabetes is estimated to cause 4.8% of all blindness. In addition, the risk of blindness from diabetes increases with the duration of diabetes. People with untreated diabetes for years are 25 times more likely to be blind than people without diabetes.

The complication of diabetes, called Diabetic retinopathy (DR), affects the blood vessels of the retina and can lead to impaired vision or blindness. With the rising prevalence of diabetes worldwide, DR has become a significant problem. Early detection, proper control of diabetes, and regular eye exams are essential to prevent vision loss.
The American diabetes association (ADA) recommends that people with diabetes have an OCT scan of their eyes every year. This is because OCT can help to detect early signs of DR with high precision. In some cases, eye care specialists may recommend more frequent OCT scans. This may be the case if the patient has advanced diabetic retinopathy or a family history of diabetic retinopathy.

AI algorithms such as Altris AI can assist in detecting the pathological signs of diabetic retinopathy or diabetic macular edema. Our web platform differentiates certain pathological signs that indicate diabetes-related diseases. Among these are:
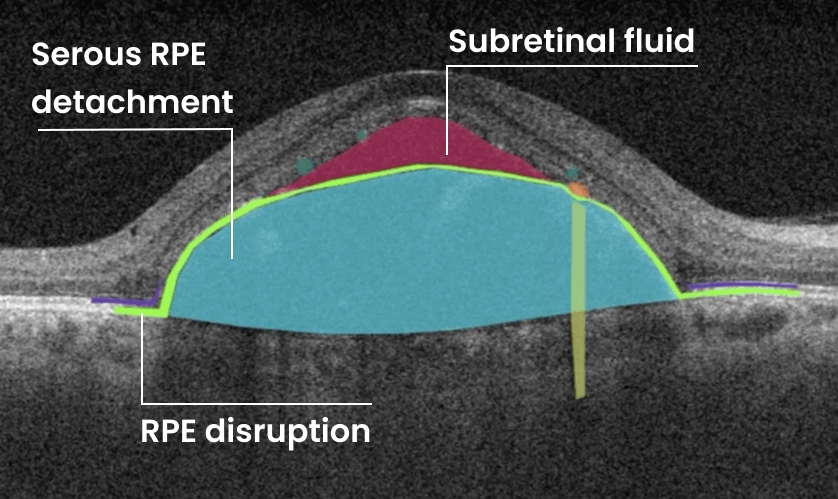
- Intraretinal fluid
- Subretinal fluid
- Hard exudates
- Hyperreflective foci
- Epiretinal fibrosis
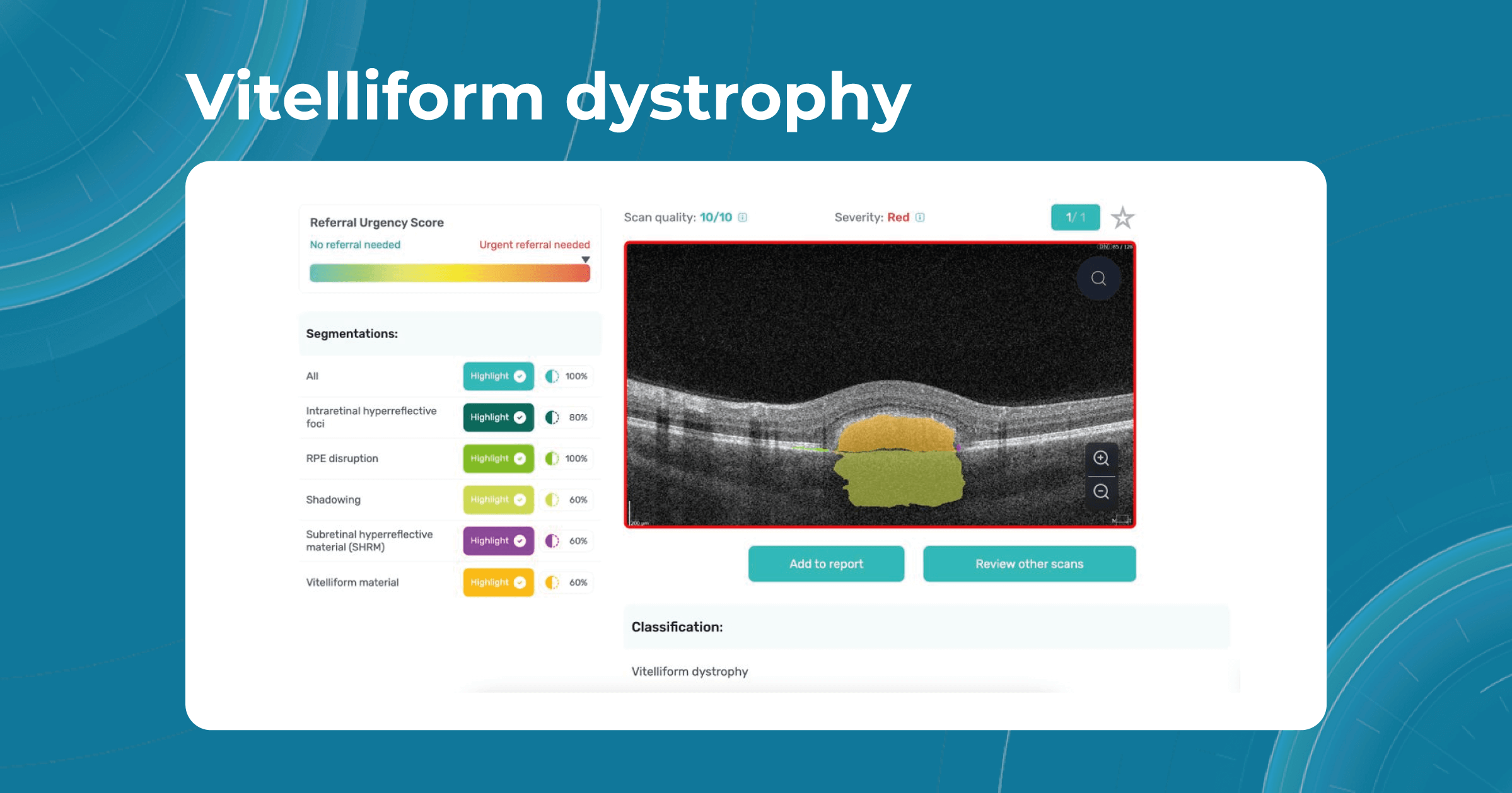
Genetic and inherited conditions: AI for visually impaired
Some patients are at a greater risk of developing visual impairment due to genetic factors or the inheritance of certain conditions. For example, retinitis pigmentosa is an inherited disease that affects the photoreceptor cells in the retina and gradually leads to night blindness and loss of peripheral vision. Genetic testing and counseling can help identify people at risk and provide early intervention.

Some genetic eye conditions, such as myopia, vitelliform dystrophy, or retinoschisis, can be detected in the early stages with the help of OCT examination and artificial intelligence systems. Altris AI platform can help eye care specialists in their daily practice and make eye care more accessible, allowing specialists to perform regular eye check-ups, and provide timely treatment of genetic conditions.

Current ways to prevent blindness with AI
As you can see, blindness risk factors encompass a wide range of conditions, pathologies, and circumstances that can significantly impact a patient’s health and increase the likelihood of severe visual impairment. Poorly managed age-related eye diseases, genetic and hereditary factors, and chronic health conditions can lead to eye-related complications, further elevating the risk of blindness.

In the following paragraphs, we will describe in detail the modern ways of using artificial intelligence to detect and prevent blindness: from AI-based retinal imaging for early detection of eye diseases to personalized treatment recommendations and remote patient monitoring.
AI for image interpretation

It is important to understand that the timely detection of eye diseases is key to the effective treatment of visual impairments. However, today we have an unfortunate tendency to diagnose severe forms of disease too late. A large-scale survey by Eyewire conducted in 2021 found that about 40% of people in the USA said they had not had an eye exam in more than a year, and 10% said they had not had one in more than five years.
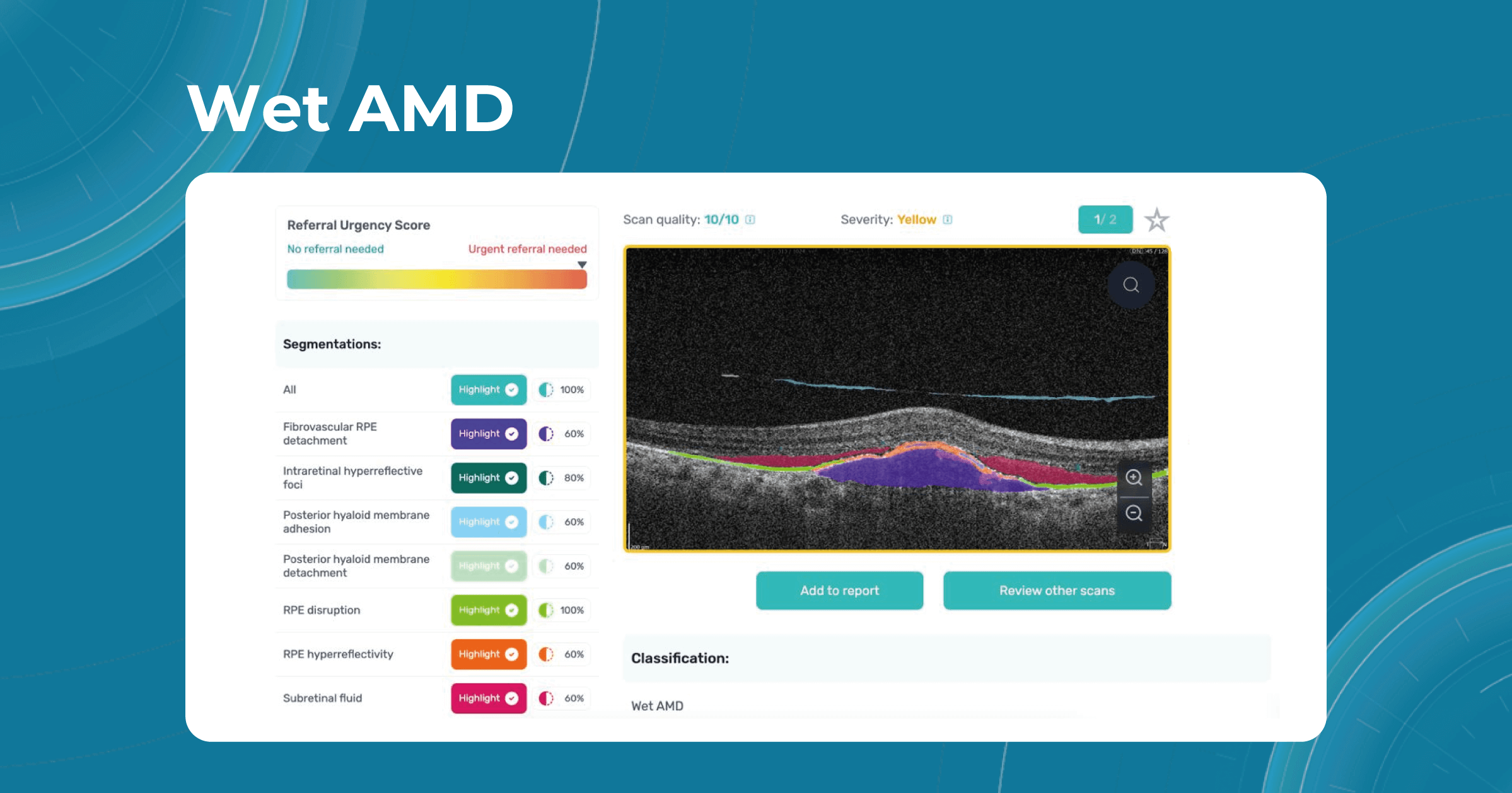
In addition, recent research by the British Journal of Ophthalmology found that 25.3% of people in Europe over the age of 60 have early signs of AMD. In the UK, about 200 people a day are affected by a severe form of AMD (wet AMD), which can cause severe blindness.
These studies show us that while eye care specialists around the world are trying to treat as many patients as possible, unfortunately, many patients are going blind due to delays in diagnosis. However, using advanced AI-based image analysis systems can speed up the detection of warning signs, allowing you to reach more patients.
One of the advantages of AI for image analysis is its assistance in decision-making. Altris AI is a great example of how an image analysis system can help prevent blindness with AI. The platform allows eye care specialists to detect 74 retina pathologies and pathological signs, including risk conditions for vision loss, like AMD, Diabetic retinopathy, Vascular diseases of the retina, and others.
Diagnosing eye disease in children

Today, one of the most important AI blindness prevention research is focused on teaching artificial intelligence algorithms to detect retinopathy in premature infants. Retinopathy of prematurity is the main cause of childhood blindness in middle-income countries. Some researches show that around 50,000 children all over the world are blind due to the disease.
Unfortunately, experts’ forecasts show that these figures are likely to grow. Retinopathy of prematurity is becoming more and more common, especially in African countries. About 30% of children born in sub-Saharan Africa have this disease and, due to late detection and insufficient attention due to the lack of eye care specialists, can also go blind.
An artificial intelligence model developed by an international team of scientists from the UK, Brazil, Egypt, and the US, with support from leading healthcare institutions, is able to identify children who are at risk of blindness if left untreated. The team of scientists hopes that this AI system will make access to screening and monitoring of young patients more affordable in many regions with limited eye care services and few qualified eye care specialists.
AI monitors for eye strain control
Another interesting application of AI to prevent blindness is eye care monitors. They are planned to be used to avoid eye strain due to prolonged computer work. Such monitors will be programmed to monitor the user’s facial expressions, blinks, and eye movements. They will also be able to assess the level of light in the room, and artificial intelligence will automatically adjust the screen brightness and image contrast.
Since a huge number of the world’s population has switched to remote work since the pandemic and spends almost all day at the computer, such AI monitors are considered a huge help for users in preventing eye diseases that can lead to visual impairment.
AI to determine better glasses or contact lenses

In the field of developing and calculating suitable lenses, there are also a number of companies that have joined the development of AI tools. AI monitors will collect important information about the patient’s eye condition, analyze it, and prescribe suitable contact lenses or glasses.
In addition, these monitors will be able to analyze the patient’s medical history, including medical images, and create the most suitable treatment strategy to maximize visual acuity.
AI for studying the human eye

Today, artificial intelligence for low vision is a promising tool for studying human eye tissue and developing new tools for diagnosing and treating eye diseases, including those that lead to vision loss. Artificial intelligence tools are used to analyze OCT images of the eye to detect changes that may indicate diseases such as diabetic retinopathy, macular degeneration, and glaucoma. AI is also used to predict the development of eye diseases based on genetic or risk factors. This is expected to help doctors identify people at risk of developing eye diseases at an early stage and prevent the progression of the disease.
Summing up
Today AI for eye diseases is already helping eye care professionals, and some companies, like Altris AI, are already using the potential of artificial intelligence to provide early detection and diagnostic advice for eye care specialists. But it’s worth noting that AI tools are not capable of coming up with innovative solutions for blindness prevention.
Only in close cooperation with eye care specialists AI blindness prevention tools can help in many ways, like early detection, providing access to medical care in underserved regions, detecting minor or rare conditions, and allowing to focus on personalized care and treatment of patients.
Disclaimer: USA FDA 510(k) Class II; Altris Image Management System (Altris IMS); AI/ML models and components intended to use for research purposes only, not for clinical diagnosis purposes.
-

5 Tips When Introducing the OCT Eye Exam to Patients
 Mark Braddon
24.07.20238 min read
Mark Braddon
24.07.20238 min readAs optometry technology evolves, many optometrists predict that utilizing OCT eye exam in practice will be vital in maximizing patient care. That is why successfully integrating an OCT device into your optometry practice workflow is instrumental to its clinical and commercial success.
Optometrists from different countries often have the same questions about how to successfully integrate an OCT device into an Optometrist Practice, regardless of practice size or experience level. How to make patients feel comfortable? How to explain the importance of regular OCT scans? Will patients understand what is an OCT scan of the eye? How do we avoid patients thinking we want to perform OCT eye exams just to earn more money? The process of introducing OCT to patients is complex and covers many areas.
If we speak to optometry practices, both those who are new to OCT and those who have had the OCT device for many years, most of them will want to improve the ROI and ensure the patients are gaining the full value of the OCT eye test. This article will show you 5 tips for successfully introducing the OCT eye exam to your patients.
Remember why you invested in the OCT technology
One may think that only novice optometrists tend to underestimate their work or do not feel confident about the value they give to patients. However, some experienced clinicians also avoid offering OCT eye tests because they think they are ‘overselling’ with additional fees for OCT, Optos, or other diagnostic exams.
That is why it is important to remember why you invested in OCT technology in the first place. In almost all cases, this is to improve the clinical standard of eye care that you offer to your patients. In fact, when I ask some optometrists if they want a member of their families to have an OCT eye exam, the answer is always ‘Yes, of course!’. So if you strongly recommend undergoing an examination to your relatives, why would you not recommend an OCT eye test for your patients?

Before a patient comes into the practice, one of the most important things you need to do is not undervalue your time, skills, and experience when charging for the additional time the OCT exam takes to interpret and discuss.
Implementing an OCT eye exam into regular practice improves clinical care and can generate a commercial benefit as well by increasing revenue through fees, patient retention, and loyalty. Moreover, word of mouth is often the most significant source of new patients for optometrists. If the patient feels you are confident in everything you do, it will make them more likely to recommend you to friends and family
Explain the importance of OCT eye exam for early detection
From the first touch point, the patient should understand that your optometric practice takes its business seriously and provides additional diagnostic examinations, such as the OCT, to improve the quality of care. The first impression of your approach is very important, so it is crucial to start introducing the technology to the potential patient from the first point of contact.
As a rule, the beginning of a patient’s introduction to the OCT eye exam starts with several touch points. Whether they make their appointment for the eye examination through your website, mobile application, in person, or by phone, the most important thing you can do is create an integrated and comfortable patient journey.

Before a patient comes into the practice, you should explain the importance of the OCT device and its benefits compared to the standard examination. Even when the patient is fully acquainted with the OCT eye exam, they may still need help understanding why this particular imaging method is necessary. The ability of OCT eye exam to detect diseases in the early stages makes this technology indispensable for optometrists and patients and this is why it is such an excellent tool for diagnosing eye diseases.
More importantly, avoid frightening patients with stories about difficult-to-treat rare pathologies. Instead of talking about the pathology consequences, say that the OCT eye exam scan provides a clear map that helps locate areas of the eye with abnormalities or early changes.
Understand the importance of a healthy-eye-as-a-baseline concept
In this section, I want to discuss the concept of a healthy eye in more detail. When a patient comes to you for an examination, it is essential to use the correct narrative that the optometrist should use when discussing the results of an OCT eye exam with patients. It is important to emphasize that we are not looking for pathology but a healthy eye.
We know that we will detect pathology in certain patients. The number of patients likely to have at least one pathology increases if you work with an older population. However, finding a healthy baseline scan is an important part of monitoring the long-term eye health of the patient.

Talking about baseline, make sure to emphasize how great it is to find a healthy eye in a patient. Explain that together you found a nice, healthy eye so you have the baseline to compare with the patient’s future scans. Emphasize that, hopefully, you will find a healthy eye at the next eye examination, but if anything does start to change, then with the help of an OCT eye exam, you will be able to detect these early and minor changes as you have the healthy baseline scan to compare to.
It is necessary to develop your patient’s understanding through appropriate teaching and discussion. Giving the value of the baseline OCT eye exam to your patients is very important. Notice the difference between “We found nothing” and “We found a healthy eye”. The first statement is negative and undermines the reason for the scanning of patients for a healthy eye baseline. Meanwhile, the second statement is positive and clearly gives your patient more value as you have found what you are looking for.
Integrate the OCT eye exam into the patient workflow
Another one of my recommendations is to call the eye examination that includes the OCT eye exam the Advanced or Comprehensive Eye Examination. It is important to make sure all the staff members use the same terminology and your message to a patient is consistent from first contact to the end of the practice visit. The eye examination without the OCT exam can be called the ‘Standard Examination’ as we are not trying to make the ‘normal’ eye examination appear below standard, what we are trying to do is explain that the practice is invested in the latest technology to offer the most advanced (or comprehensive) examination for your patients benefit.

For example, when a patient books an appointment, make sure that the support staff uses the same terminology as written throughout the website, reminder letter/email, or mobile app if you have one.
When you review the OCT images with the patient, explain that you are going to look at the OCT images of the retina, which is part of the ‘Advanced examination’. When a patient pays at the end of the customer journey, make sure that the ‘Advanced Examination’ is mentioned again. When a patient rings up or books online for the next OCT eye exam, then they will understand what the ‘Advanced examination’ means and are more likely to select this option straight away for future examinations.
Concentrate on giving more value to your patients
Review the results with the patient to give them the actual value of an OCT scan. This will allow you to establish communication with the patient and improve their perception. Give them the “theatre” around the additional diagnostic testing so they understand how it applies to them and feel valued.

Remember that your knowledge, enthusiasm, and the extent to which the patient is involved in the process directly affect the clinical and commercial success. Dedicate time to each patient, involve them in the diagnostic process, and explain the OCT scans of their eyes on the screen.
How can Altris AI help with introducing OCT Eye Exam

When talking about improving the clinical standard of care your practice offers to your patients, the Altris AI platform can also improve the standard of care you offer to your patients. The platform helps to quickly determine if the eye is healthy. If pathology is detected, then Altris AI identifies the very early, rare, or minor changes that can be the start of something more severe. Altris AI detects over 70 pathologies and pathological signs. If early pathology is identified, then the Altris AI platform can help educate the patient by clearly highlighting the areas of concern and then giving you the opportunity to discuss lifestyle changes, over-the-counter medications, or supplements, which may help the patient now rather than just monitoring until it is time to refer.
The Altris AI platform can improve the patient’s understanding of the OCT exam and add value to the Advanced Eye Examination.

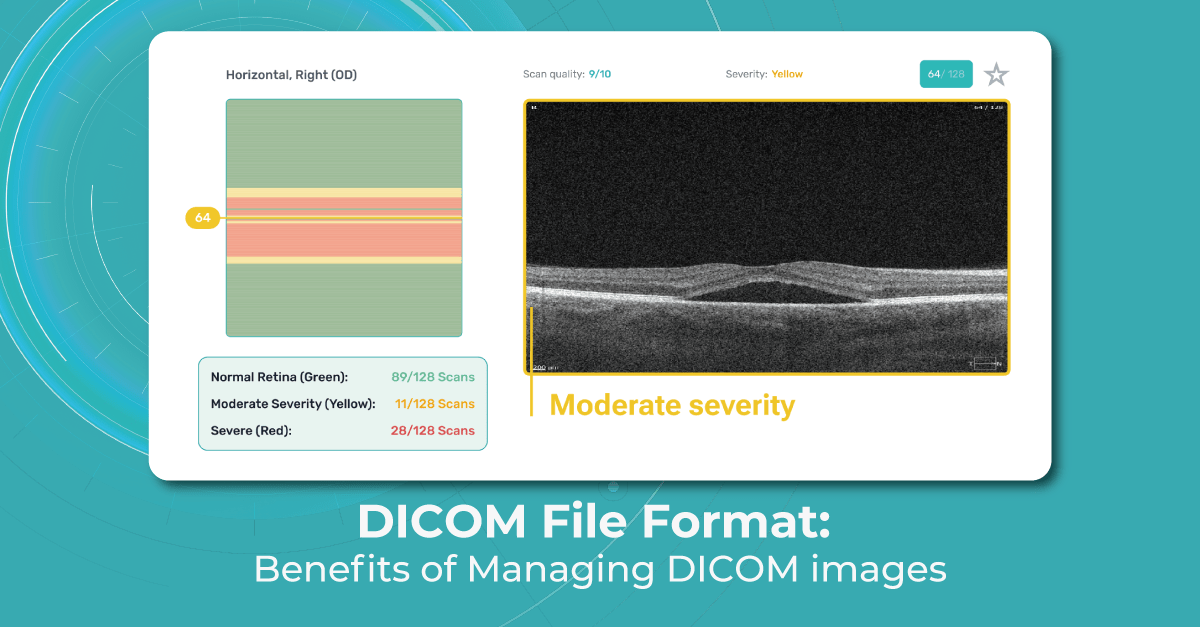
All you need to do is to upload an OCT macula exam to the platform and Altris AI will assess the exam by severity differentiating the b-scans between high, medium, and low severity levels. The segmentation/classification module will highlight pathological signs on the OCT scan one by one and give the classification/s of any pathology found to support you with the diagnosis. Meanwhile, in the Comparison module of the platform, you are able to compare the baseline scan with the current one.
Summing Up
Remember why you invested in the OCT technology in the first place — usually, this is to improve the clinical standard of care you can offer to your patients. The improvement in clinical care can also generate a commercial benefit as well by increasing revenue through OCT exam fees, patient satisfaction, patient retention and loyalty, and an increase in recommendations of friends and family.
Build a patient journey in such a way that, at each stage, they know that they have received a new, exciting, and, important part for the most comprehensive examination you offer. Remember that the more skill and enthusiasm you show, the more you can interest the patient and increase the probability that they will return for their next examination with OCT.
In addition, consider using modern AI tools to help you with decision-making. Image management systems like Altris AI can help you interpret the OCT scans faster and with more confidence. This will leave more time to add value for your patient, and integrating AI into practice can be another example of how you are investing in the latest technology to benefit your patients.
Disclaimer: USA FDA 510(k) Class II; Altris Image Management System (Altris IMS); AI/ML models and components intended to use for research purposes, not for clinical diagnosis purposes.
-

Business Case: Lux Zir and OCT Analysis in Ophthalmic Clinic
 Altris Inc.
11.07.20232 min read
Altris Inc.
11.07.20232 min readBusiness Case: Lux Zir and OCT Analysis in Ophthalmic Clinic
The Client: Lux Zir is one of the best-known ophthalmic clinics in Ukraine which provides retina diagnostics and eye treatment services. The clinic currently employs 3 full-time eye practitioners 2 general ophthalmologists and a pediatric retina expert.
The clinic normally sees between 15-20 per day with up to 10 OCT examinations performed.
The Problem:
Luxzir uses Optical Coherence Tomography as one of its core diagnostic methods because of its high level of accuracy and non-invasiveness. However, the clinic needed to solve several typical problems related to OCT.
- Some ECPs have less experience with OCT interpretation than others and this creates an inconsistent standard of care throughout the clinic.
- Some ophthalmologists come across complex OCT scans that they are unable to interpret without the help of their more experienced colleagues.
- It is difficult to maintain a high standard of care for diagnostics when the CMO is absent during the period of vacation or sick leave.
- Take out wrong and start with an inaccurate diagnosis on the basis of OCT of the patients who are referred to the clinic from other eye care centers.
The Solution:
Lux Zir Ophthalmic Clinic decided to implement the Altris AI platform as they understood how it can help resolve their problems. The results have been very positive with improvements with all issues above problems, and received very positive results.

According to Marta Shchur, Chief Medical Officer at Lux Zir clinic, the implementation of the Altris AI system improved the level of OCT diagnostics inside the clinic or if to be precise:
- OCT interpretation is now considerably faster allowing the ECPs to see 10% more patients per day.
- OCT diagnostics has become much more efficient: supported by Altris AI, ophthalmologists now have confidence when diagnosing pathologies and pathological signs, even rare ones.
- The quality of diagnostics is consistent regardless of the experience of the specialists.
Disclaimer: USA FDA 510(k) Class II; Altris Image Management System (Altris IMS); AI/ML models and components intended to use for research purposes, not for clinical diagnosis purposes.
-

Business Case: Altris for Jeff Sciberras Optometry
 Altris Inc.
10.07.20231 min read
Altris Inc.
10.07.20231 min readBusiness Case: Altris for Jeff Sciberras Optometry
The Client: Canadian Optometry Clinic
Jeff Sciberras Optometry Clinic is an established eye care facility in Mississauga, Canada. They have been recognized as the Top Choice Optometry Clinic for the past five years running in this large Canadian city.
Dr. Jeff Sciberras is proud of his high patient satisfaction rate: 92% of those surveyed would refer a friend, colleague, or family member to this establishment.
Dr. Sciberras aims to provide comprehensive eye care, with a desire to utilize leading technologies and the delivery of premium eye care products.
Recent technology investments include OCT, which allows earlier diagnosis and greater in-house management capabilities.
The Challenge: The optometry clinic has just purchased a brand new Optopol Revo OCT equipment and the support was needed in OCT scan interpretation. OCT is one of the most accurate methods of retina diagnostics however, the interpretation of OCT scans can be challenging and time-consuming, for both doctor and patient.
The Result:
Dr. Sciberras has been extremely satisfied with the support that the Altris AI platform provides:
- Increased confidence when working with the new OCT device · more profound analysis of OCT scans
- More adequate referral of complex cases.
- Scan summaries for the patient.
- Earning patient confidence and trust: The image of the innovative optometry center is enhanced to their patients and families.
- The AI Segmentation/Classification Module is invaluable for the optometry center as this module helps in the identification of 70+ pathologies and pathological signs.
The introduction of OCT with Altris AI has transformed my practice literally overnight. The integration was seamless and Altris customer support has been outstanding.
Overall, Dr. Sciberras has been impressed with the experience and support Altris AI provides and is happy to have chosen to partner with them for his leading eye care center.
- Disclaimer: USA FDA 510(k) Class II; Altris Image Management System (Altris IMS); AI/ML models and components intended to use for research purposes, not for clinical diagnosis purposes
-

DICOM Format: Benefits of Managing DICOM images
 Mark Braddon
31.05.20236 min read
Mark Braddon
31.05.20236 min readDICOM Format: Benefits of Managing DICOM images
DICOM file format (Digital Imaging and Communications in Medicine) was developed by the American College of Radiology (ACR) and the National Electrical Manufacturers Association (NEMA) as a standard for exchanging medical images and related information across different healthcare systems. It serves as a universal language for medical imaging, enabling interoperability between various imaging devices and systems. DICOM ensures that medical images can be exchanged and viewed consistently regardless of the manufacturer or modality.
DICOM image format supports a broad range of medical imaging modalities, including X-ray, MRI, OCT, ultrasound, nuclear medicine, and more. It also covers related data, such as patient information, study details, image annotations, and results
As the DICOM format continues to evolve to keep up with advancements in medical imaging technology, our article aims to raise awareness among ophthalmologists and optometrists about the DICOM file format.
What is DICOM format? You can also watch a short video about DICOM and non-DICOM file formats.
What is DICOM file format?
Image files that adhere to part 10 of the DICOM standard are commonly known as “DICOM format files” or simply “DICOM files,” and their file extension is “.dcm.” In ophthalmology, DICOM is a widely used file format for storing and transmitting medical images. DICOM files are used to store various types of ophthalmic images as well, including retinal images, optical coherence tomography (OCT) scans, visual field tests, and angiography images.
DICOM files consist of two main components: the header and the image data. The header contains metadata that describes the patient, study, series, and image acquisition parameters.

This metadata includes information such as patient demographics, image acquisition parameters (e.g., imaging modality, image orientation, pixel spacing), and any annotations or measurements made on the image. The image data itself is typically stored in a compressed format, such as JPEG or JPEG 2000, within the DICOM file.
DICOM files also support the exchange of images and associated data between different medical imaging devices and systems. This enables eye care specialists to easily share and access ophthalmic images across different platforms, such as picture archiving and communication systems (PACS), ophthalmic imaging devices, and electronic health record (EHR) systems.
By using DICOM, ophthalmologists and optometrists can efficiently store, retrieve, and analyze ophthalmic images, ensuring accurate diagnoses and effective patient care. In the next paragraphs, we will tell you more about the benefits of the DICOM file format for eye care specialists.
Benefits of DICOM format
The DICOM standard ensures interoperability between different vendors’ OCT devices and facilitates seamless data sharing and analysis. The main difference between DICOM and other image formats is that it groups information into data sets. A DICOM file consists of several tags, all packed into a single file. It stores such info as:
- demographic details about the patient
- imaging study’s acquisition parameters
- image dimensions
- matrix size
- color space
- an array of additional non-intensity information necessary for accurate image display by computers.
If you have to enter the patient’s information manually, there’s always a chance you can misspell the name or other information. However, when using a DICOM file to store patients’ information and monitor patients’ health, eye care specialists can be sure the chance of human bias is much lower.
When you work in an optometry practice or a clinic, you may spend a lot of time filling in the details every time you upload a file. And if your clinic is busy and you do 30-50 uploads daily, it could take hours. Using DICOM image format significantly speeds up the process and reduces errors.

Another benefit of the DICOM image format is that the header data information is encoded within the file so that it cannot be accidentally separated from the image data.
DICOM files can be stored in a DICOM server or transmitted between DICOM-compliant systems using the DICOM network protocol (DICOM C-STORE or DICOMweb). DICOM SR (structure reporting) allows for the structured representation of measurement data and annotations in OCT images. It enables the storage of quantitative measurements, such as retinal thickness or optic nerve parameters, as structured data within the DICOM file.
In addition, eye care specialists are able to manipulate the brightness of the image when using the DICOM viewing software. Some areas of an image can be increased or decreased for a better viewing and diagnostic experience.
Is DICOM file format popular among OCT providers?
When it comes to optical coherence tomography, many OCT device manufacturers and software providers support the DICOM standard for storing and exchanging OCT images. Some of the prominent OCT providers that offer DICOM support include:
- Heidelberg Engineering is a well-known provider of OCT devices and software solutions for ophthalmology. They offer OCT devices like the Spectralis OCT, which supports DICOM connectivity. The DICOM capabilities of their systems enable seamless integration with PACS and other healthcare systems.
- Carl Zeiss Meditec is a leading manufacturer of ophthalmic devices, including OCT systems. Their OCT devices, such as the Cirrus OCT, are DICOM-compatible, allowing for efficient storage and sharing of OCT images with other DICOM-compliant systems.
- Topcon Medical Systems is another prominent provider of OCT devices. Their OCT systems, such as the Topcon 3D OCT, support DICOM connectivity, enabling interoperability with other DICOM-enabled devices and systems.
- NIDEK offers a range of ophthalmic imaging devices, including OCT systems. Their OCT platforms, such as the NIDEK RS-3000, support DICOM, allowing for seamless integration with DICOM-compliant infrastructure, such as PACS and EHR systems.
These are just a few examples of OCT providers that support the DICOM standard. It’s important to note that DICOM support may vary among different models and versions of OCT devices from each manufacturer. We recommend you consult with the specific manufacturer or review their product documentation to confirm the DICOM capabilities of their OCT systems.
Why do we recommend using DICOM file format with Altris AI?
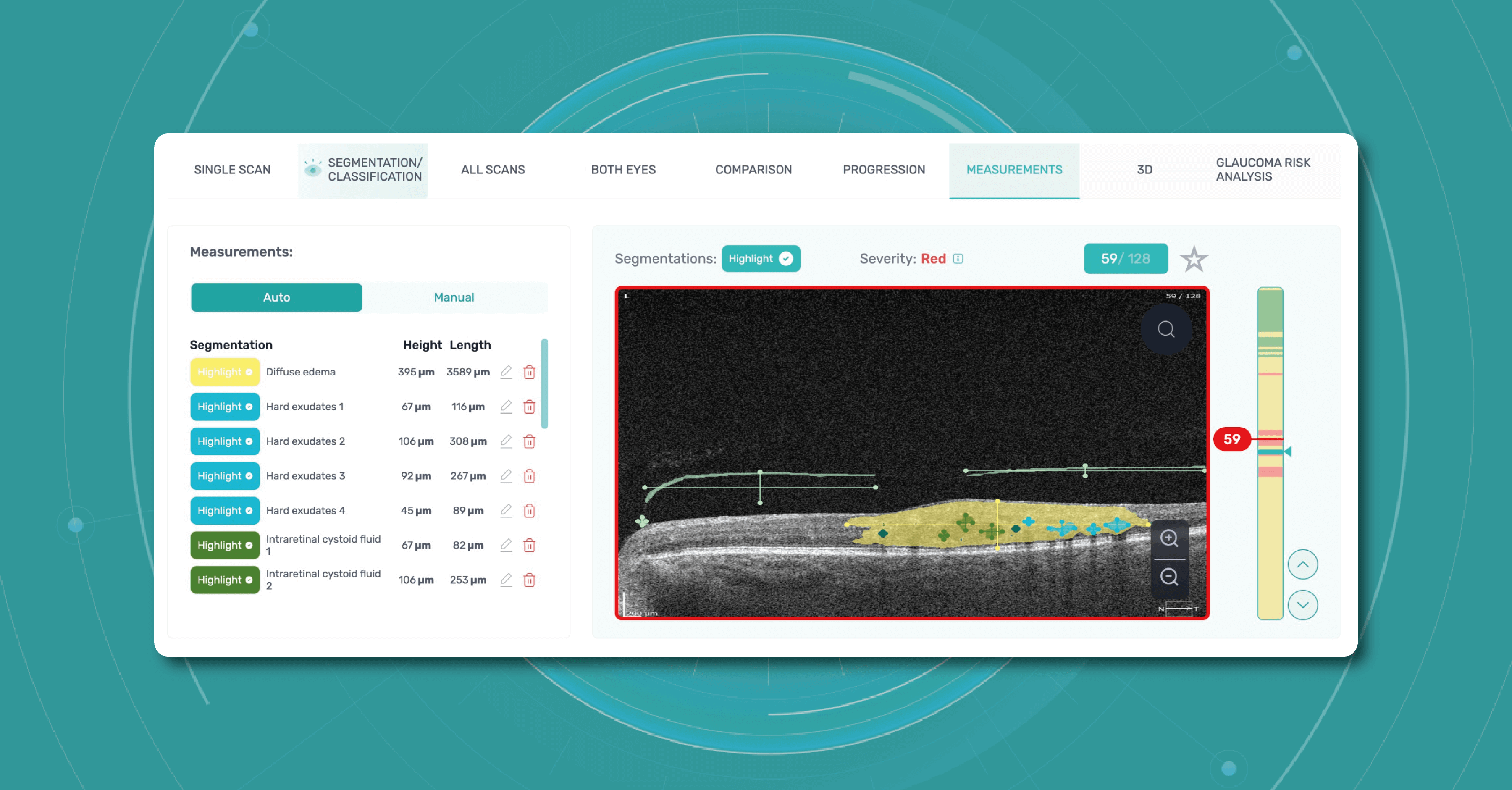
Modern DICOM viewer software extends beyond simple viewing. It can enhance image quality, generate additional data, take measurements, and more, and Altris AI is no exception. Using the DICOM image file gives you more opportunities within the platform.
Such features as
- retina layers thickness and linear measurements

- area and volume calculations

are only available when using the DICOM file format. This is because it contains the original image pixel data without modifying the study metadata. In case you upload an image, retina layers thickness won’t be available, as well as the measurements.
Another advantage of the DICOM format is that you can add patient and examination details in a few clicks by just uploading a DICOM file since this information is being pulled out automatically.

In the case of other image formats, when uploading an examination, you would have to manually fill in a bunch of information such as scan widths, eye type, etc.
Considering all mentioned above, using DICOM format files saves time, increases efficiency, and gives you more opportunities within the Altris AI platform.
Summing up
What is DICOM format? In conclusion, the DICOM file format proves to be a valuable asset for eye care specialists. Its unique characteristics, such as grouping information into data sets and incorporating standardized tags within a single file, offer many advantages.
This format ensures the preservation of accurate and comprehensive data, reducing the potential for human error and minimizing the risk of data loss or misinterpretation. The DICOM file format streamlines the archival, organization, and display of images, optimizing the workflow of eye care specialists.
By adhering to the DICOM standard, OCT devices and software solutions ensure compatibility, interoperability, and consistent data representation across different platforms. This enables efficient communication and collaboration among healthcare professionals, enhances research capabilities, and promotes the broader use and exchange of OCT imaging data.
With its widespread adoption and compatibility with various medical imaging systems, DICOM empowers ophthalmologists and optometrists to provide efficient and high-quality care while promoting seamless collaboration and knowledge sharing within the field. Ultimately, the DICOM file format plays a vital role in enhancing patient care, advancing research, and fostering innovation in the field of eye care.
FAQ
1. Why is DICOM the preferred format for ophthalmic imaging workflows?
DICOM is the industry standard because it stores both the image and full clinical metadata (patient data, scan parameters, measurements) in one structured file, enabling interoperability across devices and systems.
2. What makes DICOM better than standard image formats like JPEG or PNG in clinical use?
Unlike flat image formats, DICOM preserves diagnostic context, including acquisition settings, annotations, and structured metadata, which are essential for accurate interpretation and longitudinal tracking.
3. How does DICOM improve efficiency in AI-powered platforms like Altris AI?
DICOM allows automatic extraction of patient and scan metadata, reducing manual input and enabling faster processing, standardized workflows, and more reliable AI-based analysis of retinal images.
4. What is the key benefit of using DICOM data in AI-driven retina analysis?
It ensures full data integrity — the image pixel data and metadata remain linked, enabling precise measurements, longitudinal comparison, and more accurate AI model outputs without data loss or mismatch.
-

How 7 Leading Optometry Centers Provide Innovations in Eye Care
 Maria Martynova
08.05.20239 min read
Maria Martynova
08.05.20239 min readDisclaimer: USA FDA 510(k) Class II; Altris Image Management System (Altris IMS); AI/ML models and components intended to use for research purposes, not for clinical diagnosis purposes.
Top modern optometry centers are not afraid of embracing effective eye care innovation. Some offer home eye tests, others create mobile apps to try on frames remotely. There are optometry centers that use artificial intelligence to empower optometrists in OCT/ fundus interpretation. We’ve collected 7 optometry centers that are using technology now to win the competition.
From advanced diagnostic and treatment technologies to personalized care and patient education, these centers are transforming the way clients approach and bring innovations in eye care.
Optometry meets technology: AI, AR, mobile apps, and home eye tests
Augmented Reality (AR), mobile apps, and home eye tests are emerging trends that are changing the way people receive eye care.
- AR technology uses the camera lens on a mobile device or your PC as the method to deliver information and graphics. A user accesses an AR application, and the camera viewpoint incorporates the data directly into the perspective in real time. With AR apps for eyewear and exams, anyone can have a large selection of glasses and other services from their homes, offices, or on the go.
- Mobile apps offer a wide range of eye care services, from information on eye health and tips for maintaining healthy vision to virtual vision screenings. Moreover, mobile apps are also used to educate both young and experienced optometrists. We strongly believe that educational mobile apps inevitably become an additional efficient tool for OCT education because they are accessible and interactive.
- Another one of the innovations in eye care is Home eye tests are also often enabled by digital vision testing tools. They are becoming more and more common and offer a convenient and cost-effective way to monitor vision changes.
- As for AI use in optometry practice, it allows its users to see a broader perspective of a patient’s eye health. Incorporating AI streamlines billing procedures, expands the input of electronic health records (EHRs), optimizes claims management, and improves cash flow. AI technology can also be used in cooperation with AR assisting in the glasses selection.
Although these innovations in optometry and ophthalmology provide more comprehensive access to eye care and improve patient engagement, many optometry practices are still hesitating to add such innovations to their routine. That is why we prepared the info about 7 famous optometry practices that are already using innovations in eye care.
Warby Parker

Warby Parker started its way in 2010 when the founders of the company were students. One of them lost his glasses during a tourist trip. The cost of replacing them was so high that he spent his first semester of graduate school without them. That is why the company’s mission is to provide affordable, high-quality eyewear to consumers, while also addressing the issue of access to vision care.
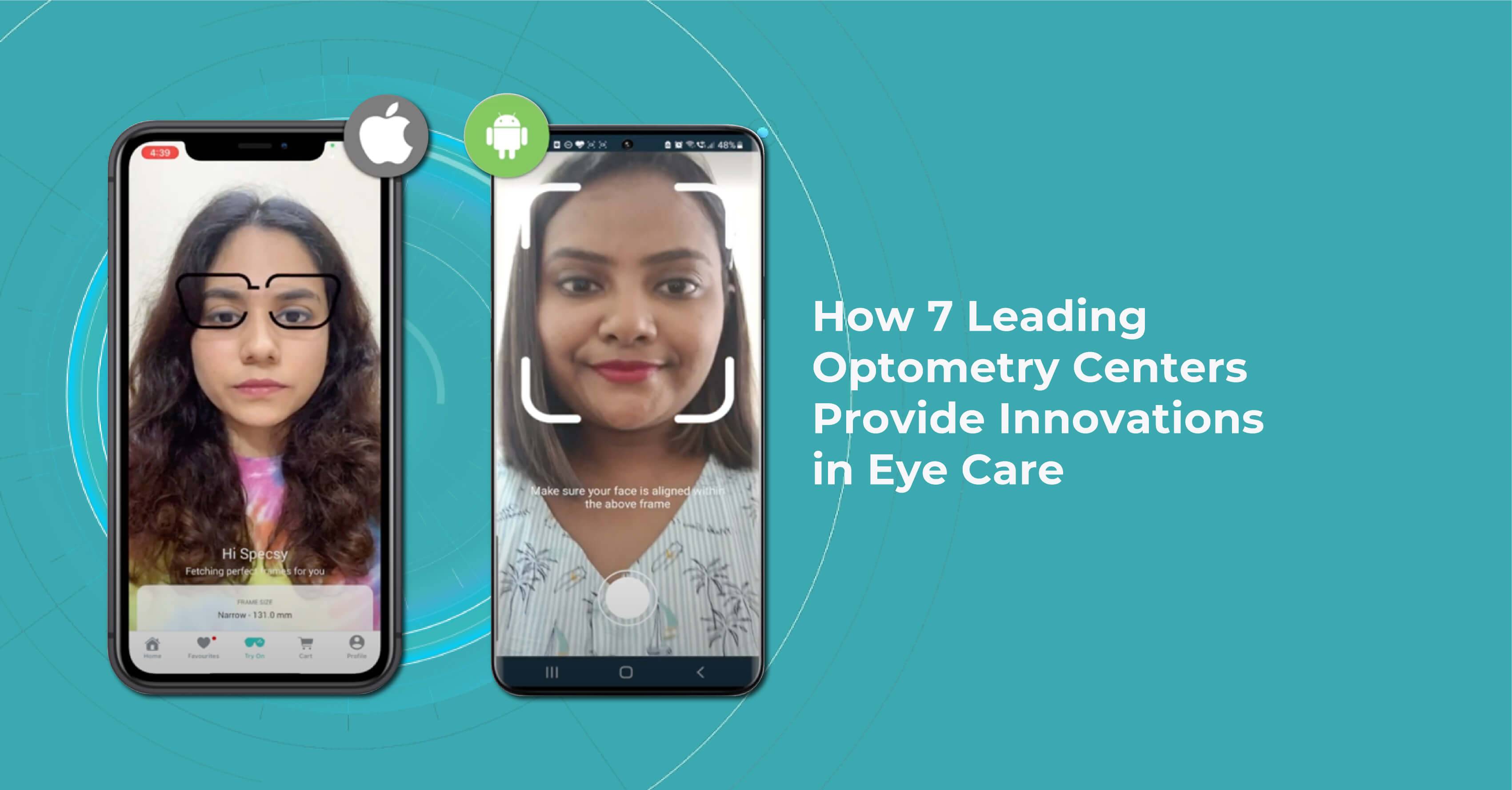
One of Warby Parker’s unique innovations in eye care is its Home Virtual Try-On program, which allows customers to try on up to five frames at home for free before making a purchase. This program makes it easier for customers to find the perfect pair of glasses and eliminates the need for them to go to a physical store to try on frames.

Warby Parker also offers an online eye exam called the Virtual Vision Test. It is designed to provide customers with a convenient and affordable way to obtain a prescription for glasses or contacts from the comfort of their own homes.
The Virtual Vision Test is a telemedicine service that uses technology to allow customers to take an eye exam using their computer or smartphone. The test is not meant to replace a comprehensive eye exam performed by an eye doctor, but rather to provide a convenient option for those who need a prescription renewal or have mild refractive errors.
After completing the test, the results are reviewed by a licensed ophthalmologist or optometrist, who will issue a prescription if appropriate. The customer can then use the prescription to purchase glasses or contacts from Warby Parker or any other provider.
Lenskart

Lenskart is a fast-growing company of innovations in eye care in India focused on making eyewear more affordable for everyone. To achieve this goal, the company has developed a number of innovative technologies and business models, including a mobile app that allows customers to try on frames virtually and a home vision testing service that allows to check their prescriptions from the comfort of their own home.
One special feature of the Lenskart app is the “3D Try-On” feature, which uses 3D imaging technology to create a model of the customer’s face and allows them to try on different frames virtually. This feature helps get a better sense of how a particular frame will look on a customer’s face before making a purchase.

Another one of Lenskart’s innovations in eye care is the Home eye test, designed to provide people with a convenient and affordable way to obtain a prescription for glasses or contact lenses. To take the Lenskart Home Eye Test, customers must first book an appointment on the company’s website or mobile app.
The eye test includes a visual acuity test, a color vision test, and a refractive error test. The optometrist will also check the customer’s eye health and recommend any necessary follow-up exams or treatments. After the test, the optometrist will provide a prescription, which the customer can use to purchase glasses or contacts from Lenskart or any other provider.
SmartBuyGlasses

SmartBuyGlasses is an online eyewear retailer that was founded in 2006. The company is headquartered in Hong Kong, but it operates in more than 20 countries worldwide. Company’s Virtual Try-On feature is available on the website and allows customers to upload a photo of themselves and try on glasses virtually using augmented reality.
After the website generates a 3D model of the customer’s face, they can adjust the position and size of the glasses to get a better sense of how they will look on their faces. The virtual try-on innovations in eye care also allow to share images of themselves wearing the glasses with their friends and family to get feedback on which pair looks best on them.

Another eye care innovation of SmartBuyGlasses is a Lens scanner app that uses advanced technology to scan the user’s current eyeglasses lenses and analyze the prescription, allowing to order a new pair of glasses online without visiting an eye doctor.
The app works by instructing the user to place their current eyeglasses on a flat surface and position their smartphone camera above the lenses. The app then captures a series of images and uses advanced algorithms to analyze the curvature, thickness, and other factors of the lenses to determine the prescription.
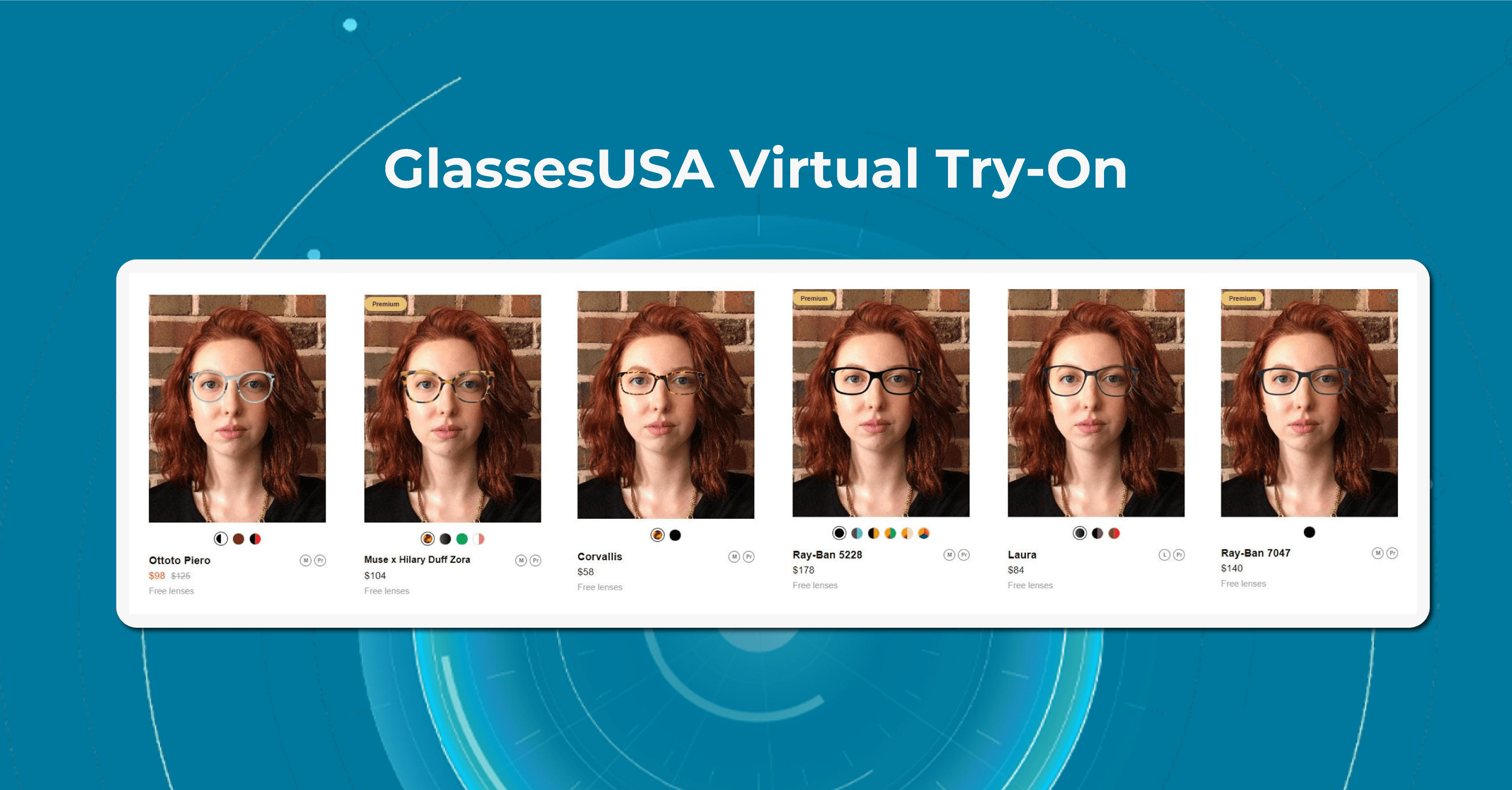
GlassesUSA

GlassesUSA is an innovative and socially responsible eyewear retailer that is committed to providing quality products and services to its customers. With its focus on technology, sustainability, and social impact, GlassesUSA has become a popular choice for customers in the United States and around the world.
One of the innovations in eye care of GlassesUSA that is worth paying attention to is a Prescription Scanner app. The app works by guiding the user through a series of steps to scan their face and eyes using their smartphone camera. It uses advanced algorithms to analyze the user’s facial features and measure the distance between their pupils, which is a crucial factor in determining the correct prescription for eyeglasses.

Once the scanning process is complete, the GlassesUSA app provides the user with their personalized prescription and recommendations. The app also offers a Virtual Try-On feature that allows users to see how different frames will look on their faces before making a purchase.
Another feature is a Find-your-Frame Quiz on the website. The quiz consists of a series of questions that ask users about their face shape, personal style, and preferences for eyeglass frames, such as color, material, and shape. Based on the user’s responses, the specially designed program generates a personalized selection of eyeglasses frames that are recommended for their face shape and style preferences.
Zenni Optical

Zenni Optical offers a wide range of eyewear products, including prescription glasses, sunglasses, and sports eyewear. The company offers glasses at significantly lower prices than traditional brick-and-mortar stores, which has made it a popular choice for customers.
Company’s Virtual Try-On feature uses advanced AR technology to create a 3D model of the user’s face, allowing them to see how different frames will fit and look on them.

To use the Virtual Try-On innovations in eye care, users simply need to upload a photo of themselves or use their computer or smartphone camera to take a live video. This feature then maps the user’s facial features and displays a selection of eyeglasses frames that can be tried on virtually. Users can then select different frames to see how they look from different angles, and can even compare different frames side-by-side.
The Zenni Optical Virtual Try-On is a convenient and easy-to-use tool for anyone in the market for a new pair of glasses. It allows users to see how different frames will look on their faces without the need to visit a physical store or try on multiple pairs of glasses.
VSP Global

VSP Global is a leading eyewear company that was founded in 1955 by a group of optometrists who wanted to provide affordable eye care. Today, VSP Global is a major player in the optometric industry and offers its customers a wide range of services and products.
The company works with a network of over 40,000 eye doctors and optometrists to provide affordable and accessible eye care to its customers. VSP Global also offers other eye care services, such as telehealth consultations, on-site eye exams for businesses and schools, and a mobile eye clinic that serves underserved communities.

AI for OCT
FDA-cleared system for your business
And as every company from this article, VSP Global has a strong focus on technology and innovations in eye care. The company has developed a number of proprietary technologies, including an AI-powered platform called Eyeconic that helps customers find the right eyewear.
Eyeconic uses machine learning algorithms to analyze a customer’s facial features and suggest frames that would fit their face shape and size. VSP Global has also developed a mobile app called myVSP that allows customers to manage their vision benefits, find an eye doctor, and order contact lenses online.
iSight+

Another AI-oriented optometry center is iSight+, located in Hong Kong. iSight+ is an excellent example of how an optometric eye care center didn’t hesitate and chose to provide innovations in eye care and a more in-depth examination of the macula.
Andy Meau. Optometrist, the owner of ISight+ Optometric Eye Care center:
“Altris AI will be a great tool in helping to monitor patients with existing macular diseases. I am also honored to be the first EPC in Hong Kong to provide this service.”
In addition, the eye care center is also equipped with advanced optometric technologies, digital photography systems, and optical coherence tomography (OCT), which helps to provide the highest quality eye examination.
Summing Up
Optometry centers can significantly benefit from incorporating innovations in eye care, such as augmented reality, artificial intelligence, and mobile apps, into their practice. These technologies enhance the patient experience, improve diagnostic accuracy, and streamline clinical workflows.
Moreover, the use of innovative technology can help optometry centers stay competitive in a rapidly evolving healthcare landscape. Patients are increasingly tech-savvy and expect healthcare providers to offer convenient, digital solutions that meet their needs. By embracing innovative technologies, optometry centers can attract new patients and retain existing ones, while also increasing operational efficiency and reducing costs.
Of course, there may be concerns about the cost and complexity of integrating new technologies into an optometry practice. However, the benefits of doing so can far outweigh these potential challenges. With careful planning and implementation, optometry centers can successfully leverage AR, AI, and other innovations in eye care to enhance patient care, improve clinical outcomes, and thrive in a rapidly changing healthcare environment.